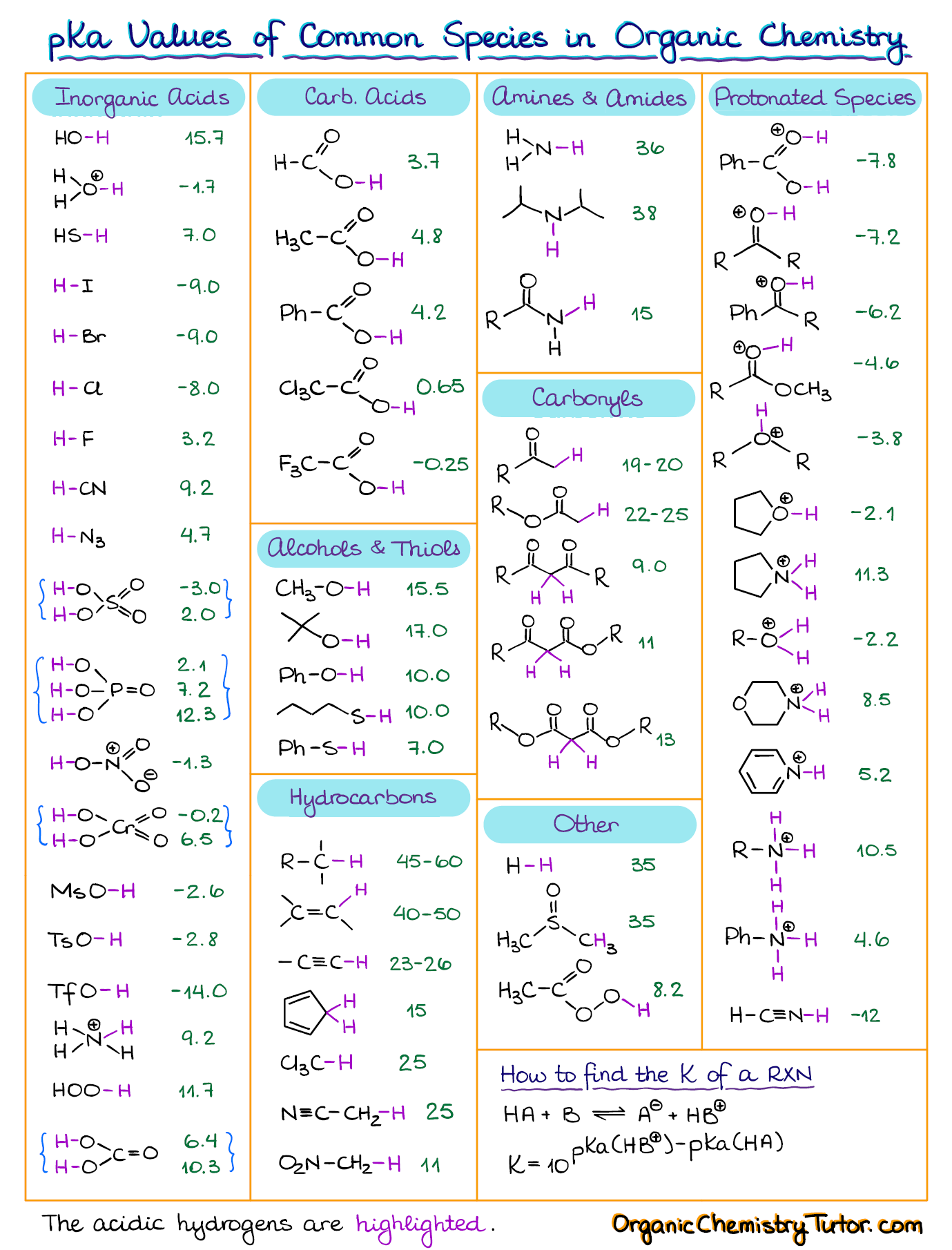
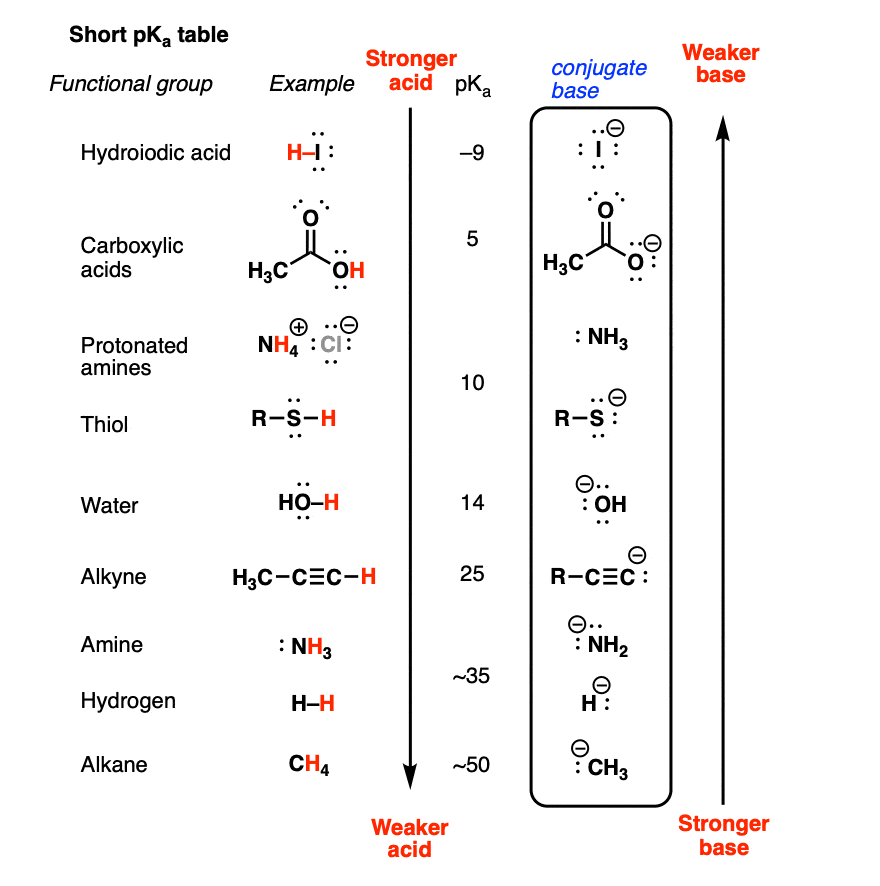
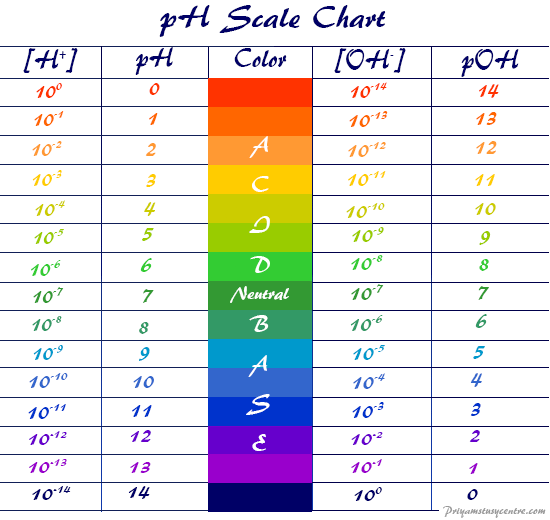
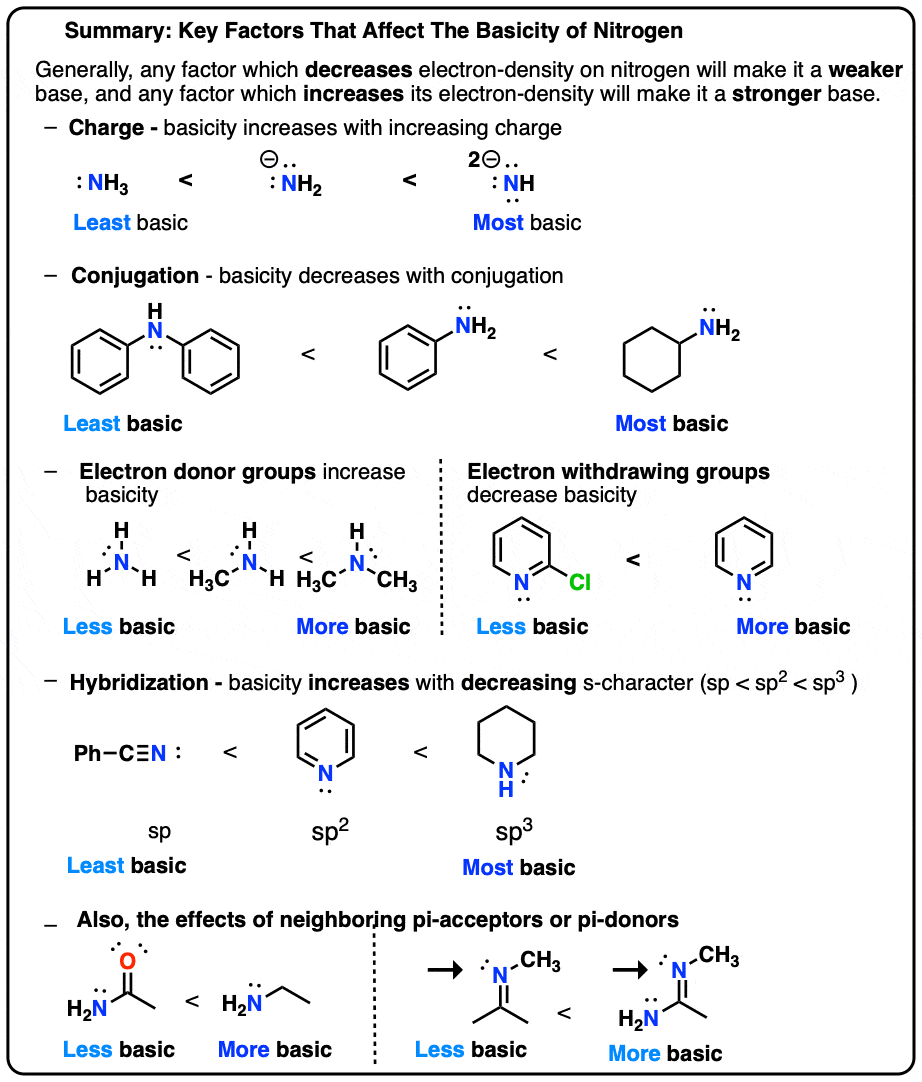
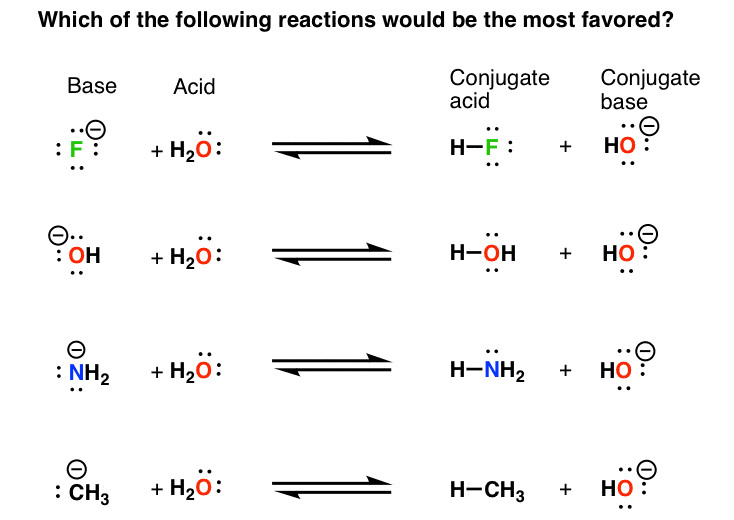
Basicity Chart
Basicity Chart - 6 realise that there is a notable drop in basicity from nitrogen to phosphorus and then a slow and continuous further diminishing. I know that to compare the basic strengths, we need to find the stability of their. Acidity and basicity depend on the interaction between the acid/base and the solvent. While steric hindrance will decrease the rate solvent access, it more importantly also. Thus, the order of basicity of aliphatic amines should be: The notable drop is due to the difference of the. Greater is the stability of the substituted. The issue is that basicity has a couple of meanings and you have to be able to tell from the context what it means. Now i have a problem with the first line. Well, i know that b(oh)x3 b (o h) x 3 is a weak. Now i have a problem with the first line. While steric hindrance will decrease the rate solvent access, it more importantly also. I need to find the decreasing basicity of the following compounds(i, ii, iii, iv, respectively): Acidity and basicity depend on the interaction between the acid/base and the solvent. Now i end up thinking basicity and reducing character are the same thing. Order of basicity for arylamines and ammonia in gas phase ask question asked 3 years, 8 months ago modified 3 years, 5 months ago Primary > secondary > tertiary. The order of acidity and basicity is based on countless experiments that establish. Thus, the order of basicity of aliphatic amines should be: Basicity decreases with increasing s character basicity decreases with increasing stability, due to resonance/ delocalization (taken from this webpage). I know that to compare the basic strengths, we need to find the stability of their. Basicity is how easily can the compound give up hydrogen atoms/electrons. Thus, the order of basicity of aliphatic amines should be: Primary > secondary > tertiary. The issue is that basicity has a couple of meanings and you have to be able to tell. Order of basicity for arylamines and ammonia in gas phase ask question asked 3 years, 8 months ago modified 3 years, 5 months ago Acidity and basicity are experimentally determined. I need to find the decreasing basicity of the following compounds(i, ii, iii, iv, respectively): 6 realise that there is a notable drop in basicity from nitrogen to phosphorus and. I need to find the decreasing basicity of the following compounds(i, ii, iii, iv, respectively): While steric hindrance will decrease the rate solvent access, it more importantly also. Acidity and basicity are experimentally determined. Basicity decreases with increasing s character basicity decreases with increasing stability, due to resonance/ delocalization (taken from this webpage). I know that to compare the basic. Now i have a problem with the first line. My book defines the basicity of the acid as the number of hx+ h x + ions furnished by one mole of the acid in solution. Now i end up thinking basicity and reducing character are the same thing. The first is logical, and is used to describe how basic a. Order of basicity for arylamines and ammonia in gas phase ask question asked 3 years, 8 months ago modified 3 years, 5 months ago Now i have a problem with the first line. Basicity decreases with increasing s character basicity decreases with increasing stability, due to resonance/ delocalization (taken from this webpage). The notable drop is due to the difference. The order of acidity and basicity is based on countless experiments that establish. Basicity is how easily can the compound give up hydrogen atoms/electrons. The notable drop is due to the difference of the. Well, i know that b(oh)x3 b (o h) x 3 is a weak. My book defines the basicity of the acid as the number of hx+. Acidity and basicity are experimentally determined. I need to find the decreasing basicity of the following compounds(i, ii, iii, iv, respectively): Order of basicity for arylamines and ammonia in gas phase ask question asked 3 years, 8 months ago modified 3 years, 5 months ago Greater is the stability of the substituted. 6 realise that there is a notable drop. Now i have a problem with the first line. The first is logical, and is used to describe how basic a base is. Thus, the order of basicity of aliphatic amines should be: The issue is that basicity has a couple of meanings and you have to be able to tell from the context what it means. I know that. Primary > secondary > tertiary. The first is logical, and is used to describe how basic a base is. Basicity is how easily can the compound give up hydrogen atoms/electrons. Acidity and basicity depend on the interaction between the acid/base and the solvent. Thus, the order of basicity of aliphatic amines should be: The order of acidity and basicity is based on countless experiments that establish. Acidity and basicity depend on the interaction between the acid/base and the solvent. The notable drop is due to the difference of the. Greater is the stability of the substituted. The first is logical, and is used to describe how basic a base is. Now i have a problem with the first line. The issue is that basicity has a couple of meanings and you have to be able to tell from the context what it means. 6 realise that there is a notable drop in basicity from nitrogen to phosphorus and then a slow and continuous further diminishing. The first is logical, and is used to describe how basic a base is. Acidity and basicity are experimentally determined. Greater is the stability of the substituted. The order of acidity and basicity is based on countless experiments that establish. Acidity and basicity depend on the interaction between the acid/base and the solvent. I know that to compare the basic strengths, we need to find the stability of their. While steric hindrance will decrease the rate solvent access, it more importantly also. Basicity is how easily can the compound give up hydrogen atoms/electrons. Primary > secondary > tertiary. My book defines the basicity of the acid as the number of hx+ h x + ions furnished by one mole of the acid in solution. I need to find the decreasing basicity of the following compounds(i, ii, iii, iv, respectively): Well, i know that b(oh)x3 b (o h) x 3 is a weak. Basicity decreases with increasing s character basicity decreases with increasing stability, due to resonance/ delocalization (taken from this webpage).AcidBase Equilibrium Part 1 How to Use the pKa Table — Organic Chemistry Tutor
Acids And Bases List Strength
Acidity, Basicity, and Stability The Relationship YouTube
Acids and Bases
pH Scale pOH Scale Definition, Range, Chart, Measurement
Secondary Amines Examples at David Masterson blog
Acidity and Basicity Constants Acidity and basicity consta… Flickr Photo Sharing!
Understanding basicity in organic chemistry — Master Organic Chemistry
Ionization Constants of BH Onium Acids Name of Base (B) Formula of B K (BH ) pK
Order of Basicity Acid and Base Strengths YouTube
Thus, The Order Of Basicity Of Aliphatic Amines Should Be:
And That Bihx3 B I H X 3 Should Have.
Now I End Up Thinking Basicity And Reducing Character Are The Same Thing.
Order Of Basicity For Arylamines And Ammonia In Gas Phase Ask Question Asked 3 Years, 8 Months Ago Modified 3 Years, 5 Months Ago
Related Post: