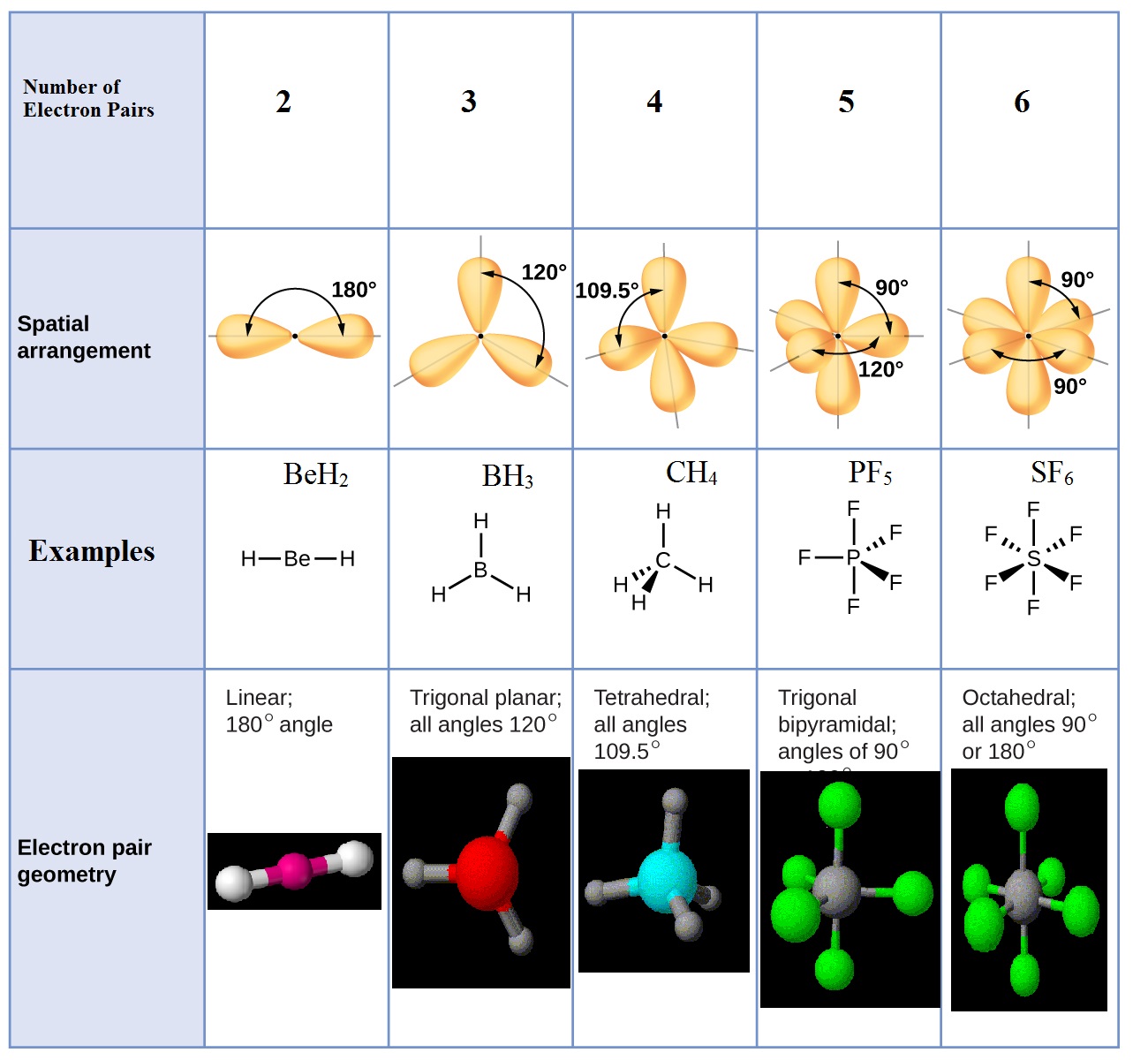
Electron And Molecular Geometry Chart
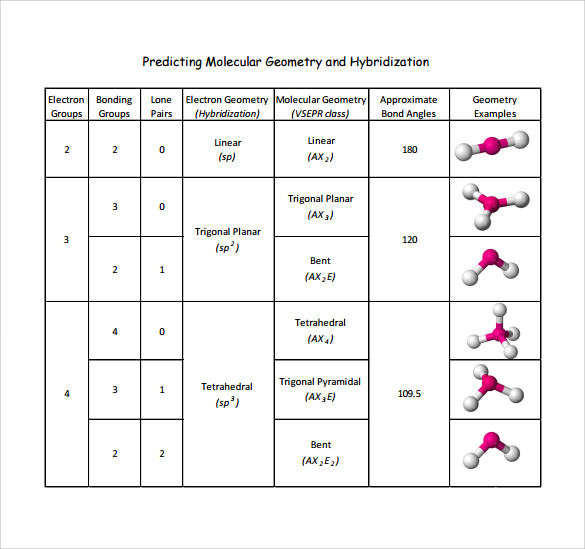
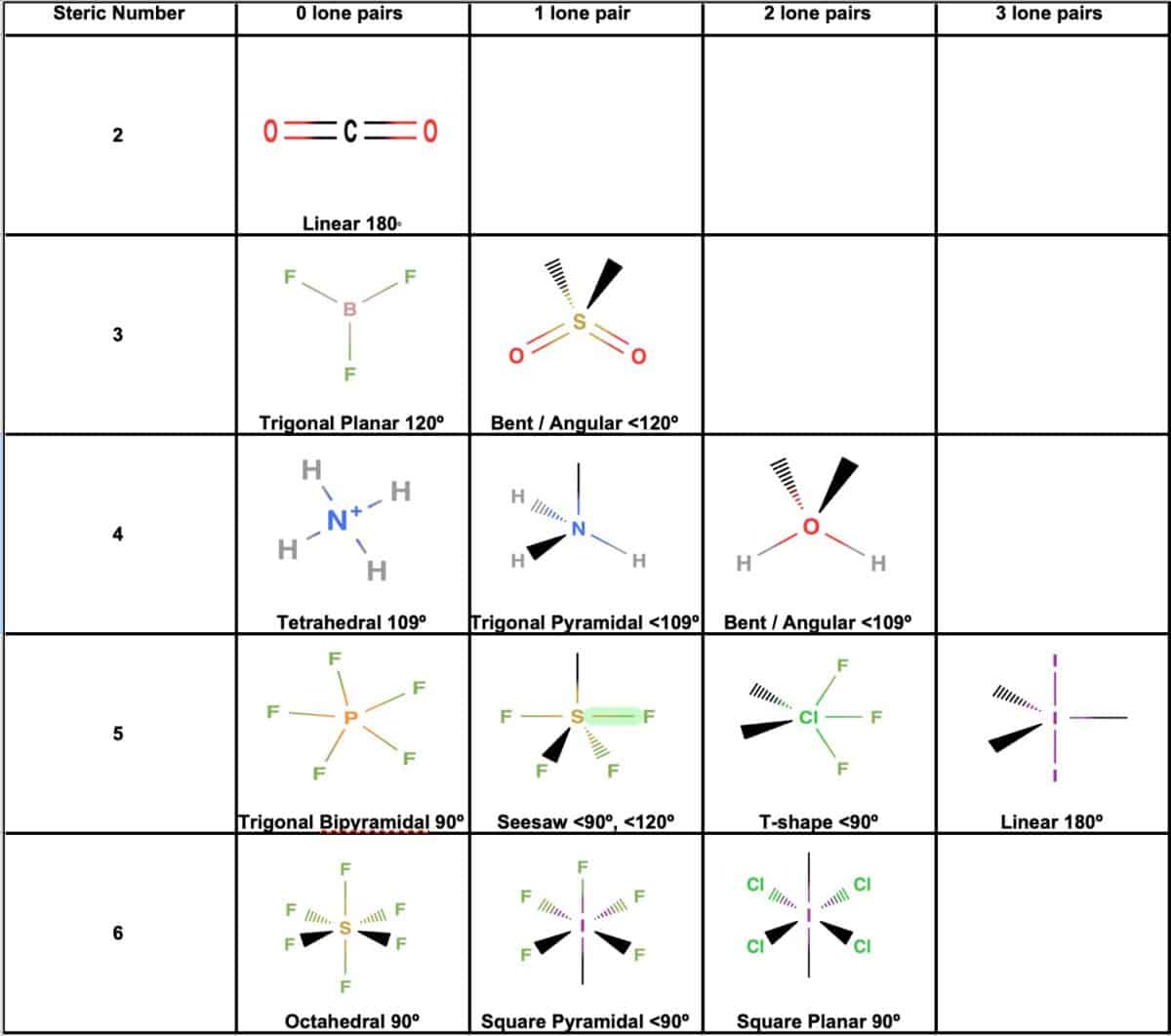
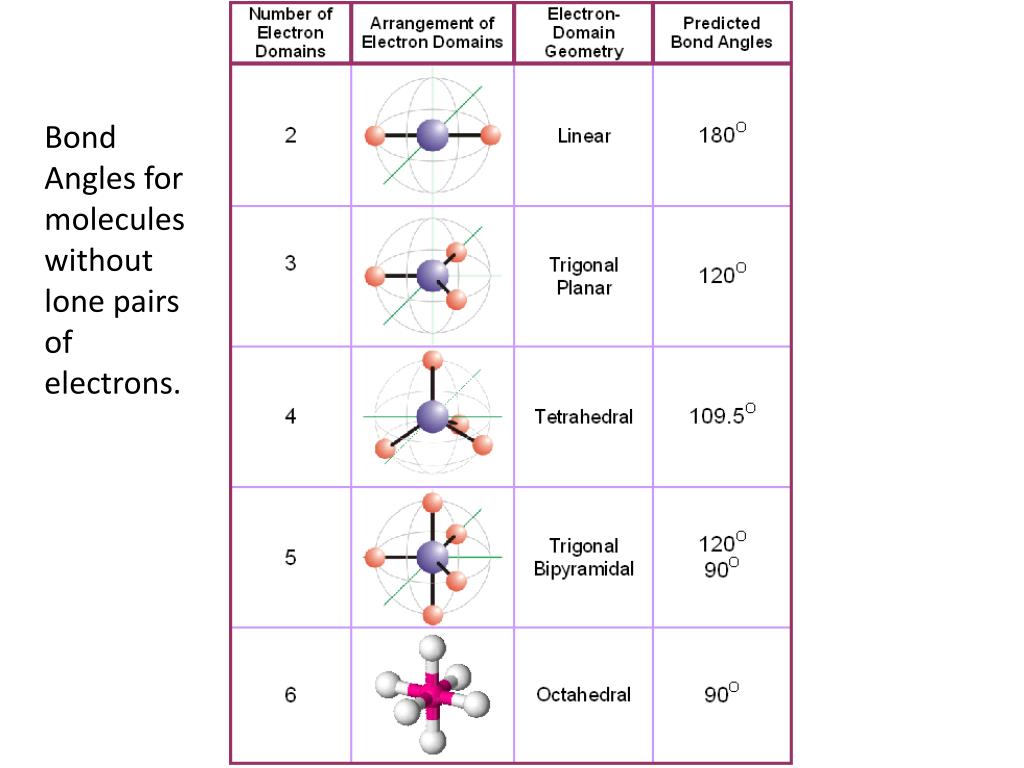
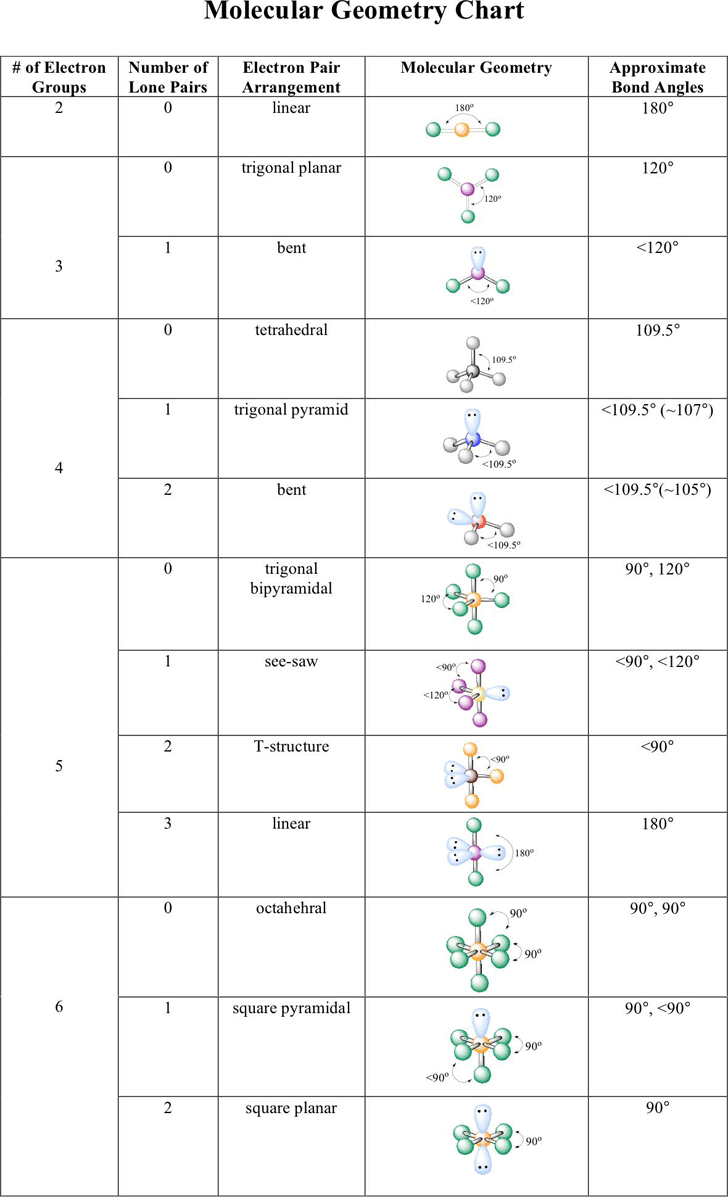
Electron And Molecular Geometry Chart - We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by focusing only on the number of electron pairs around the central atom, ignoring all other valence. We have also included some study guides to. You will need to know the electron pair geometries and molecular geometries. Lone pairs influence the molecular geometry, and so in this section we will look at. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Check out a table of molecular geometries with examples and diagrams. The table below gives the electron pair geometries and molecular geometries for different molecules and ions. What are the different shapes of molecules. Join us as we define this subject, go over some examples, and list the different structures you will find in an electron and molecular geometry chart. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. We have also included some study guides to. This vsepr chart shows you all of the common vsepr geometries, organized by the steric number and how many lone electron pairs they have. Check out a table of molecular geometries with examples and diagrams. Join us as we define this subject, go over some examples, and list the different structures you will find in an electron and molecular geometry chart. What are the different shapes of molecules. The geometrical arrangements of the atoms are referred to as molecular geometry. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Lone pairs influence the molecular geometry, and so in this section we will look at. The molecular geometry differs from the electron geometry when a molecule contains one or more lone pairs. The molecular geometry differs from the electron geometry when a molecule contains one or more lone pairs. We have also included some study guides to. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. The steric. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. We have also included some study guides to. You can manipulate the number of single, double, or triple bonds and lone pairs that are. We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by. Lone pairs influence the molecular geometry, and so in this section we will look at. You will need to know the electron pair geometries and molecular geometries. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. You can manipulate the number of single, double, or triple bonds and lone pairs that are. Check out a. What are the different shapes of molecules. The molecular geometry differs from the electron geometry when a molecule contains one or more lone pairs. We have also included some study guides to. We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by focusing only on the number of electron pairs around the central. The table below gives the electron pair geometries and molecular geometries for different molecules and ions. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. This vsepr chart shows you all of the common vsepr geometries, organized by the steric number and how many lone electron pairs they have. You will need to know the. We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by focusing only on the number of electron pairs around the central atom, ignoring all other valence. You can manipulate the number of single, double, or triple bonds and lone pairs that are. You will need to know the electron pair geometries and molecular. What are the different shapes of molecules. Lone pairs influence the molecular geometry, and so in this section we will look at. The steric number is how many atoms are bonded. You can manipulate the number of single, double, or triple bonds and lone pairs that are. The geometrical arrangements of the atoms are referred to as molecular geometry. This vsepr chart shows you all of the common vsepr geometries, organized by the steric number and how many lone electron pairs they have. The steric number is how many atoms are bonded. The table below gives the electron pair geometries and molecular geometries for different molecules and ions. You can manipulate the number of single, double, or triple bonds. Check out a table of molecular geometries with examples and diagrams. What are the different shapes of molecules. The steric number is how many atoms are bonded. We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by focusing only on the number of electron pairs around the central atom, ignoring all other valence.. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. Lone pairs influence the molecular geometry, and so in this section we will look at. We have also included some study guides to. The geometrical arrangements of the atoms are referred to as molecular geometry. What are the different shapes of. The table below gives the electron pair geometries and molecular geometries for different molecules and ions. Join us as we define this subject, go over some examples, and list the different structures you will find in an electron and molecular geometry chart. The steric number is how many atoms are bonded. This vsepr chart shows you all of the common vsepr geometries, organized by the steric number and how many lone electron pairs they have. You will need to know the electron pair geometries and molecular geometries. The molecular geometry differs from the electron geometry when a molecule contains one or more lone pairs. What are the different shapes of molecules. You can manipulate the number of single, double, or triple bonds and lone pairs that are. We can use the vsepr model to predict the geometry of most polyatomic molecules and ions by focusing only on the number of electron pairs around the central atom, ignoring all other valence. Check out a table of molecular geometries with examples and diagrams. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Lone pairs influence the molecular geometry, and so in this section we will look at.Molecular Geometry Boundless Chemistry
FREE 8+ Sample Molecular Geometry Chart Templates in PDF MS Word
Electron Geometry Chart Chemistry molecular geometry electron geometries chart shapes vsepr
Electron and molecular geometry chart examples heryhd
electrongeometrychartmoleculargeometrychartexample1 electron geometry chart Molecular
Electron geometry chart and examples numbersaad
Free Molecular Geometry Chart PDF 603KB 1 Page(s)
Electron pair geometry and molecular geometry chart volfplus
Electron Domain and Molecular Geometry Chart
Electron Geometry Chart Chemistry molecular geometry electron geometries chart shapes vsepr
We Have Also Included Some Study Guides To.
If There Are No Lone Pairs And All Orbitals Are Bonding, Then The Molecular Geometry Is The Electronic Geometry.
The Geometrical Arrangements Of The Atoms Are Referred To As Molecular Geometry.
Related Post: