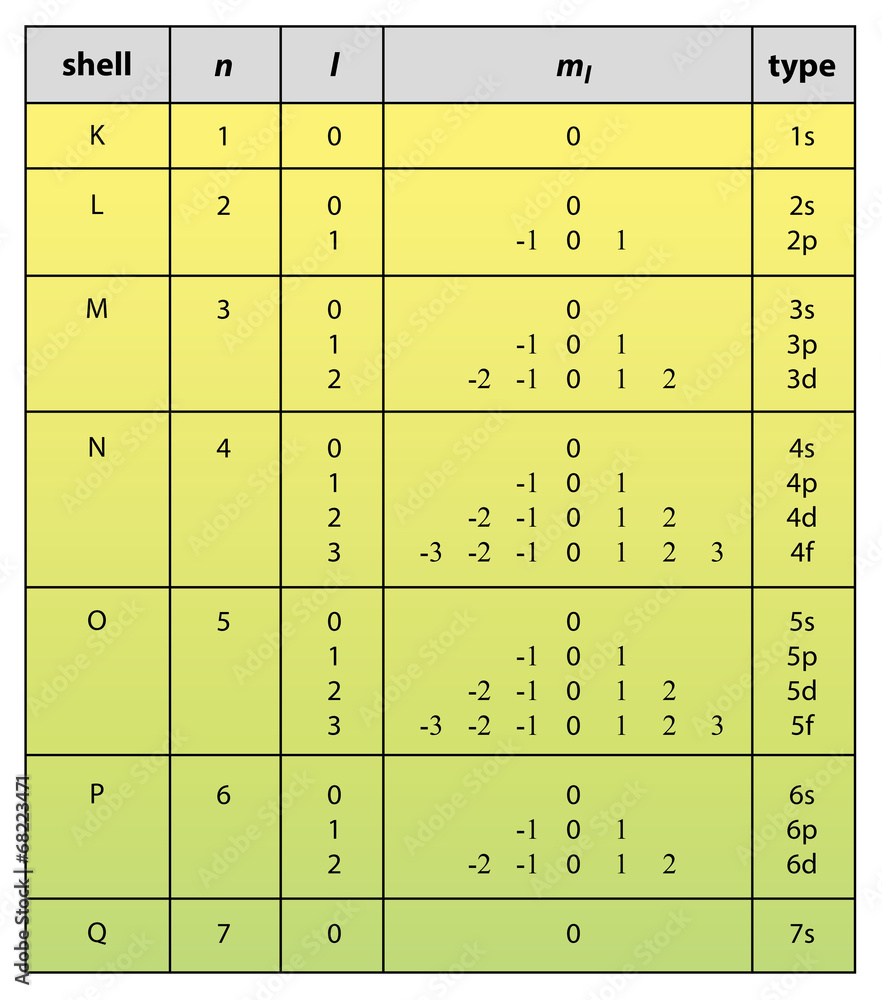
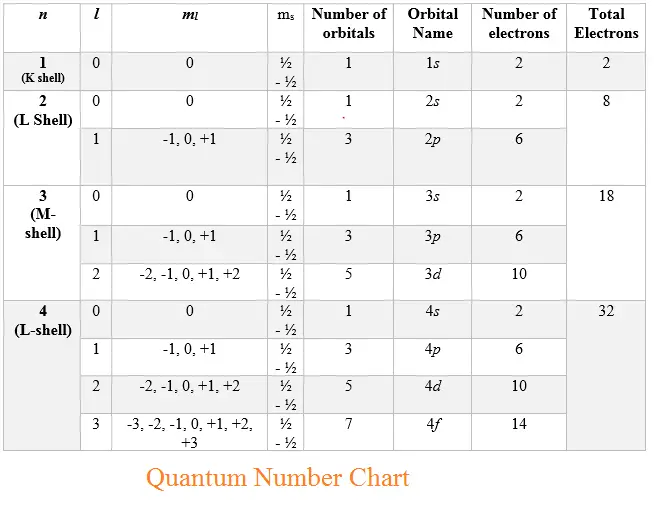
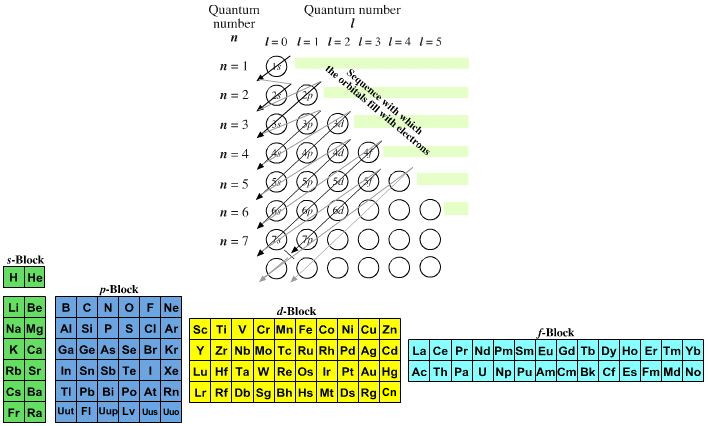
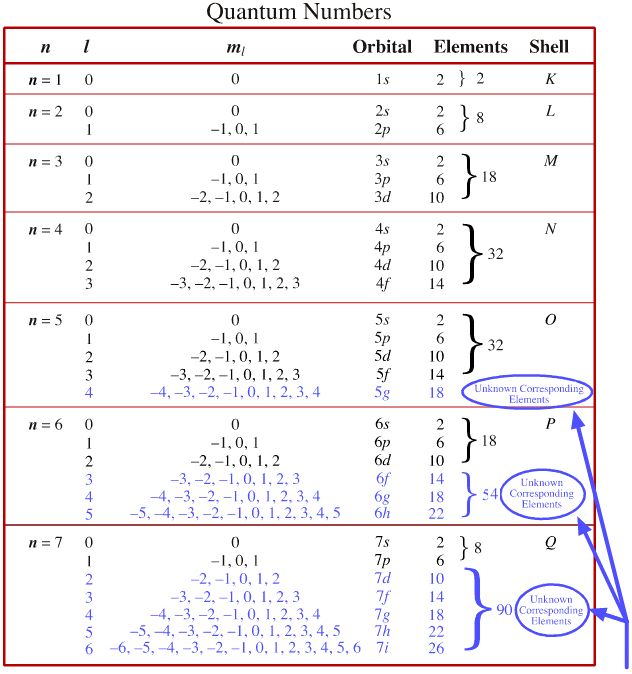
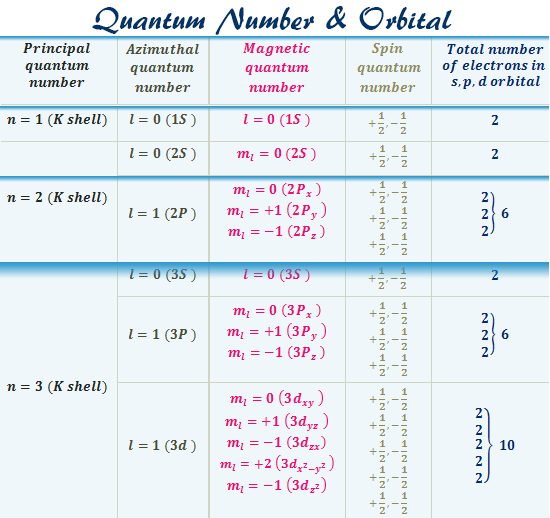
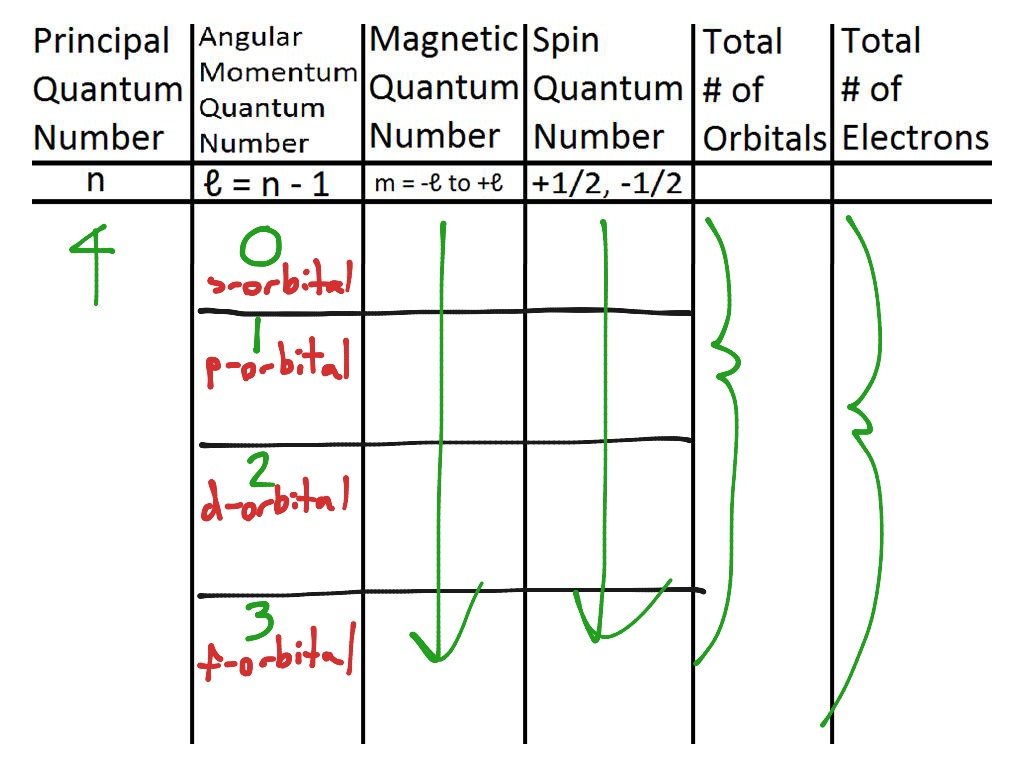
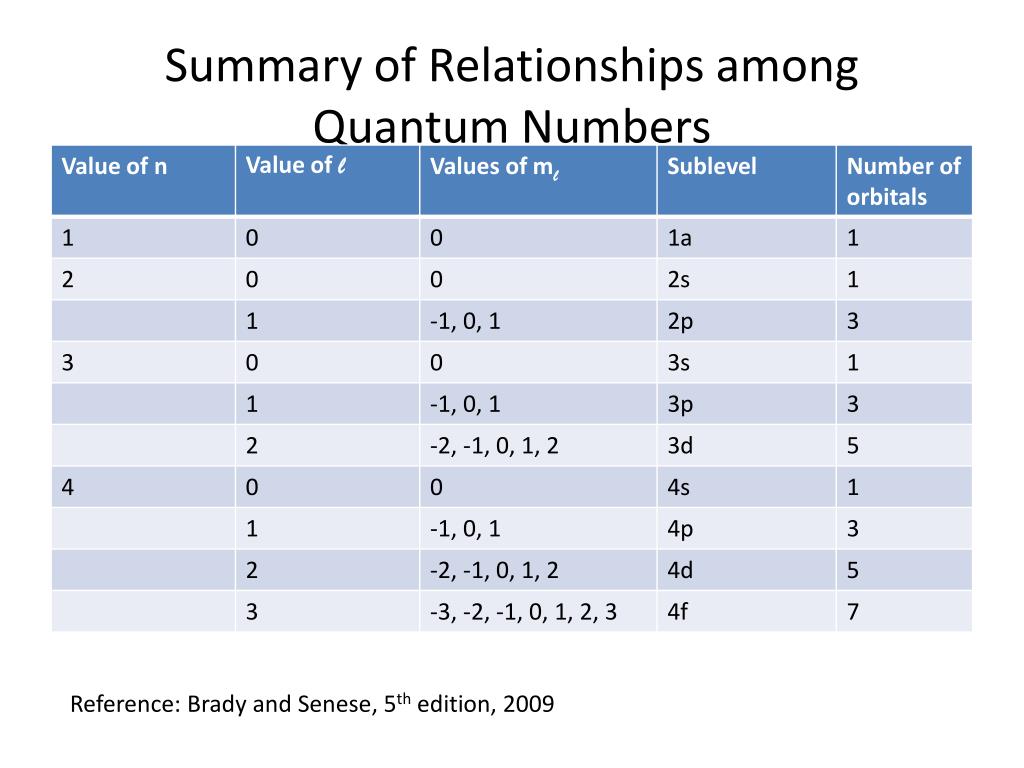
Quantum Number Chart
Quantum Number Chart - The principal quantum number #n# = 5 and the azimuthal quantum number #l# = 1 specify a 5p orbital. Prime numbers are divisible only by 1 and itself, i.e. As you know, four quantum numbers are. The bohr's atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the orbit. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can accommodate eight electrons and the 2p subshell can hold two electrons in. It is not limited to the first row. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose. I assume you are asking the question in context of quantum or statistical mechanics. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. This assumption is flawed and was further. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. As you know, four quantum numbers are. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose. The principal quantum number #n# = 5 and the azimuthal quantum number #l# = 1 specify a 5p orbital. Prime numbers are divisible only by 1 and itself, i.e. Prime numbers aren't divisible by anything except 1 and itself, composite numbers are all real numbers that aren't prime. This assumption is flawed and was further. Many transition metals have variable oxidation states. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can accommodate eight electrons and the 2p subshell can hold two electrons in. I assume you are asking the question in context of quantum or statistical mechanics. Prime numbers aren't divisible by anything except 1 and itself, composite numbers are all real numbers that aren't prime. The bohr's atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the orbit. Many transition metals have variable oxidation states. I assume you are asking the question in context of quantum. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. This assumption is flawed and was further. The principal quantum number #n# = 5 and the azimuthal quantum number #l# = 1 specify a 5p orbital. I assume you are asking the question in context of quantum or statistical mechanics. Many transition. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose. Prime numbers are divisible only by 1 and itself, i.e. The bohr's atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the. Many transition metals have variable oxidation states. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can accommodate eight electrons and the 2p subshell can hold two electrons in. When dealing with percents the word of means times or to multiply. So, for a system you can have certain observables,. The bohr's atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the orbit. This assumption is flawed and was further. When dealing with percents the word of means times or to multiply. Many transition metals have variable oxidation states. Prime numbers are divisible only by 1 and itself, i.e. Many transition metals have variable oxidation states. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose. Prime numbers aren't divisible by anything except. Many transition metals have variable oxidation states. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. As you know, four quantum numbers are. It is not limited to the first row. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can. Many transition metals have variable oxidation states. Prime numbers are divisible only by 1 and itself, i.e. The bohr's atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the orbit. As you know, four quantum numbers are. I assume you are asking the question in context of quantum or statistical. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose. Many transition metals have variable oxidation states. This assumption is flawed and was further. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100.. Prime numbers aren't divisible by anything except 1 and itself, composite numbers are all real numbers that aren't prime. When dealing with percents the word of means times or to multiply. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can accommodate eight electrons and the 2p subshell can hold. So, for a system you can have certain observables, for example considering a single particle a system, it's position. Prime numbers aren't divisible by anything except 1 and itself, composite numbers are all real numbers that aren't prime. It is not limited to the first row. As you know, four quantum numbers are. The principal quantum number #n# = 5 and the azimuthal quantum number #l# = 1 specify a 5p orbital. I assume you are asking the question in context of quantum or statistical mechanics. Put another way, the possible values of the four quantum numbers account for the fact that the second shell can accommodate eight electrons and the 2p subshell can hold two electrons in. Percent or % means out of 100 or per 100, therefore 5% can be written as 5 100. This assumption is flawed and was further. Many transition metals have variable oxidation states. According to the quantum theory, the vacuum contains virtual particles which are in a continuous state of fluctuation.casimir realised that between two plates, only those virtual photons whose.Principal Quantum Number Chart
Quantum Numbers Chart
Quantum Number Definition Types Chart And Quiz
Quantum Number Definition Types Chart And Quiz
Quantum Numbers Chart physicscatalyst's Blog
Principal Quantum Number Chart
*CHEMISTRY MATRICULATION* QUANTUM NUMBERS
Quantum Number Orbitals Diagram, Definition, Chart, Shape
Quantum Numbers Chart Chemistry
PPT Principle Quantum Numbers PowerPoint Presentation, free download ID5519904
When Dealing With Percents The Word Of Means Times Or To Multiply.
Prime Numbers Are Divisible Only By 1 And Itself, I.e.
The Bohr's Atomic Model Although Assumes That The Energy Of The Electron Depends Only On The Principle Quantum Number (N) Of The Orbit.
Related Post: