Solvent Solubility Chart
Solvent Solubility Chart - The solvent is the substance that dissolves the solute and the component of a chemical solution present in the greatest amount. It is the substance in which the solute is dissolved. Something that provides a solution. Polar solvents (e.g., water) favour formation of ion s; A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Solvent, substance, ordinarily a liquid, in which other materials dissolve to form a solution. Something that eliminates or attenuates something. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. While most common solvent are liquids, a. A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Solvent, substance, ordinarily a liquid, in which other materials dissolve to form a solution. Essential traits include its dissolving power, homogeneity, and its role as a reaction medium. The molecules of the solvent work to put the. Water is a solvent for polar molecules, and the most common solvent used by living things; A solvent is a substance that dissolves a solute to form a homogeneous solution. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. The solvent is the substance that dissolves the solute and the component of a chemical solution present in the greatest amount. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. Polar solvents (e.g., water) favour formation of ion s; It is the substance in which the solute is dissolved. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. A solvent is a substance that dissolves a. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. Usually, a solvent is a liquid. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. A usually liquid substance capable of dissolving or dispersing one or more other substances. Solvent, substance, ordinarily a liquid, in which other materials dissolve. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. A solvent is, by definition, any substance that will take other things (aka ‘solutes’) into solution. A solvent is a substance that dissolves a solute to form a homogeneous solution. A solvent is a substance that becomes a solution by dissolving a. Polar solvents (e.g., water) favour formation of ion s; A solvent is a substance that can dissolve an insoluble solute and create a solution. It is the substance in which the solute is dissolved. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. A solvent is, by definition, any substance that will. Essential traits include its dissolving power, homogeneity, and its role as a reaction medium. A solvent is a substance that can dissolve an insoluble solute and create a solution. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. Something that eliminates or attenuates something. A solvent is, by definition, any substance that will. Essential traits include its dissolving power, homogeneity, and its role as a reaction medium. The solvent is the substance that dissolves the solute and the component of a chemical solution present in the greatest amount. Water is a solvent for polar molecules, and the most common solvent used by living things; A solvent is usually a liquid but can also. Usually, a solvent is a liquid. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. A solvent is usually a liquid, but can also be a solid or gas. A solvent is a substance that dissolves a solute to. A solvent can be solid, liquid or gas. While most common solvent are liquids, a. A solvent is, by definition, any substance that will take other things (aka ‘solutes’) into solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. The molecules of the solvent work to put the. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Something that provides a solution. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. The solvent is the. Something that eliminates or attenuates something. The molecules of the solvent work to put the. Usually, a solvent is a liquid. A solvent is, by definition, any substance that will take other things (aka ‘solutes’) into solution. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. A solvent is usually a liquid, but can also be a solid or gas. Usually, a solvent is a liquid. Essential traits include its dissolving power, homogeneity, and its role as a reaction medium. Water is a solvent for polar molecules, and the most common solvent used by living things; While most common solvent are liquids, a. A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is a substance that dissolves a solute to form a homogeneous solution. A solvent is the component of a solution that is present in the greatest amount. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. Something that eliminates or attenuates something. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. It is the substance in which the solute is dissolved. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. A solvent is, by definition, any substance that will take other things (aka ‘solutes’) into solution. A solvent can be solid, liquid or gas. Polar solvents (e.g., water) favour formation of ion s;Solvent Solubility Chart
Solvent Solubility Chart
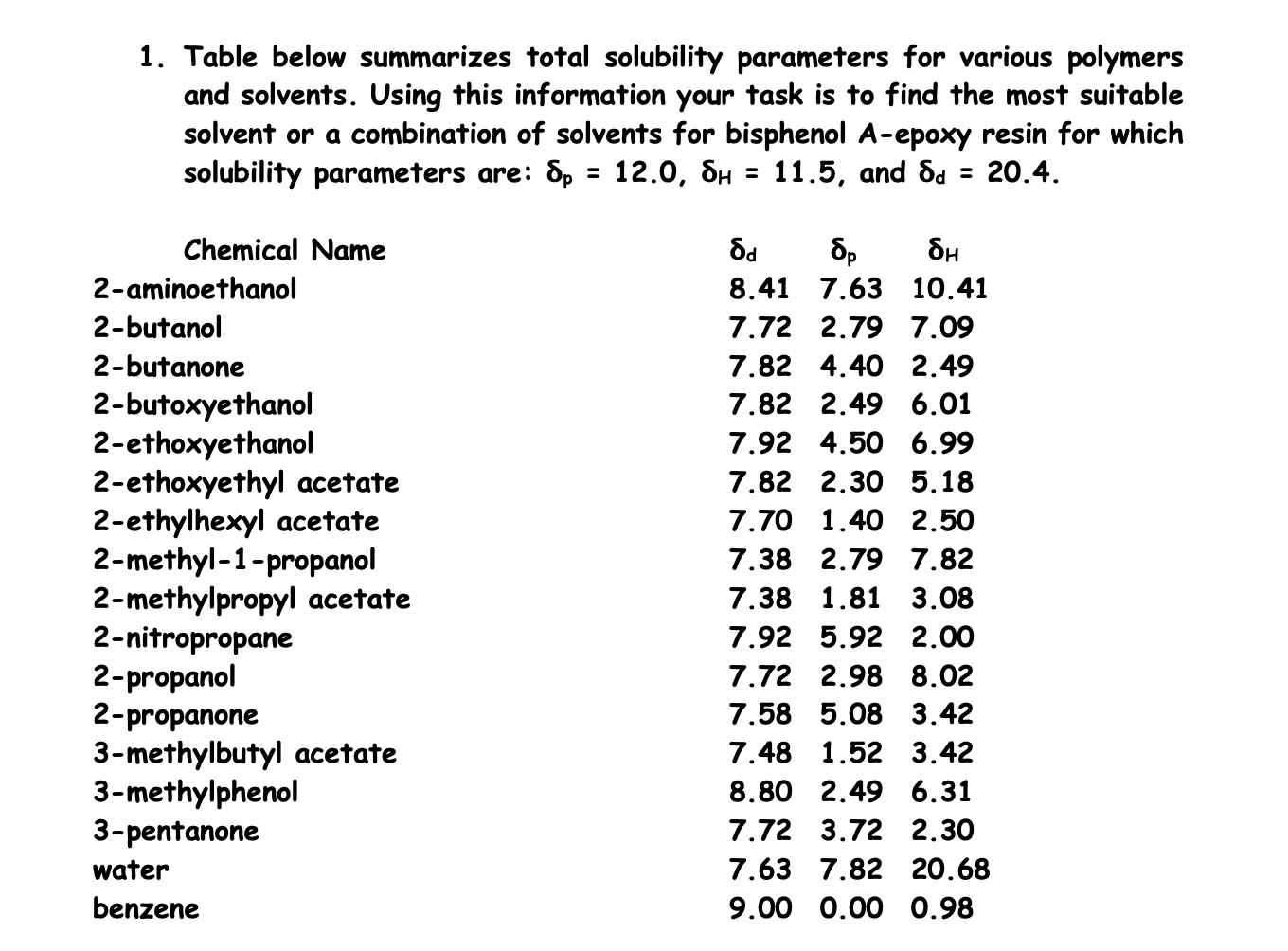
Solvent Solubility Table
Solubility Table
Solvent Solubility Chart
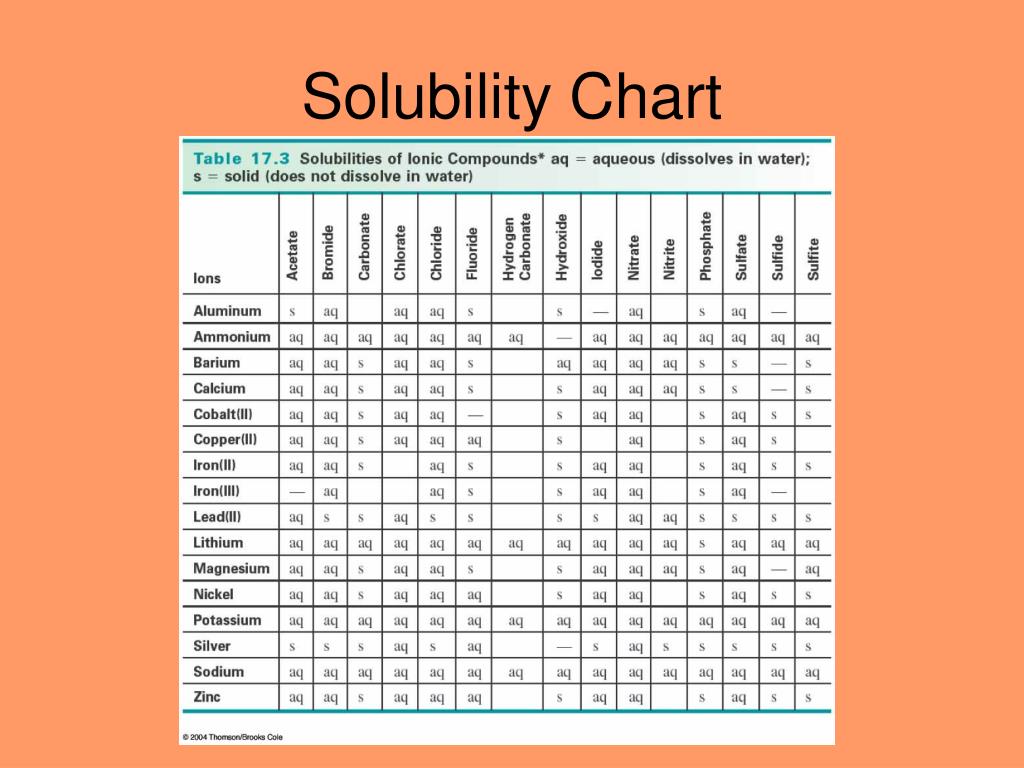
Solubility Rules Chart.png
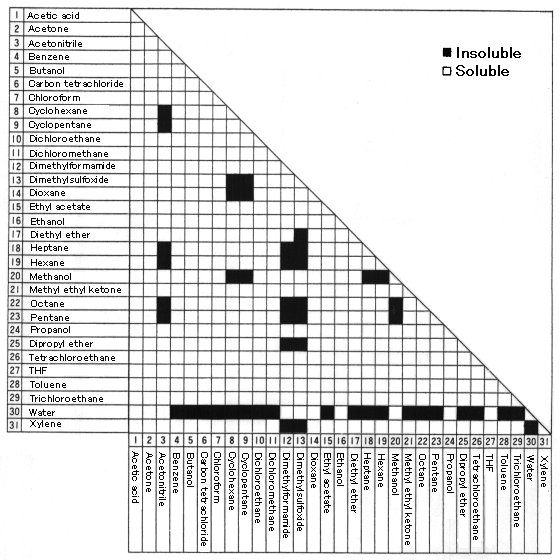
Solvent Solubility Chart
Solvent Solubility Table
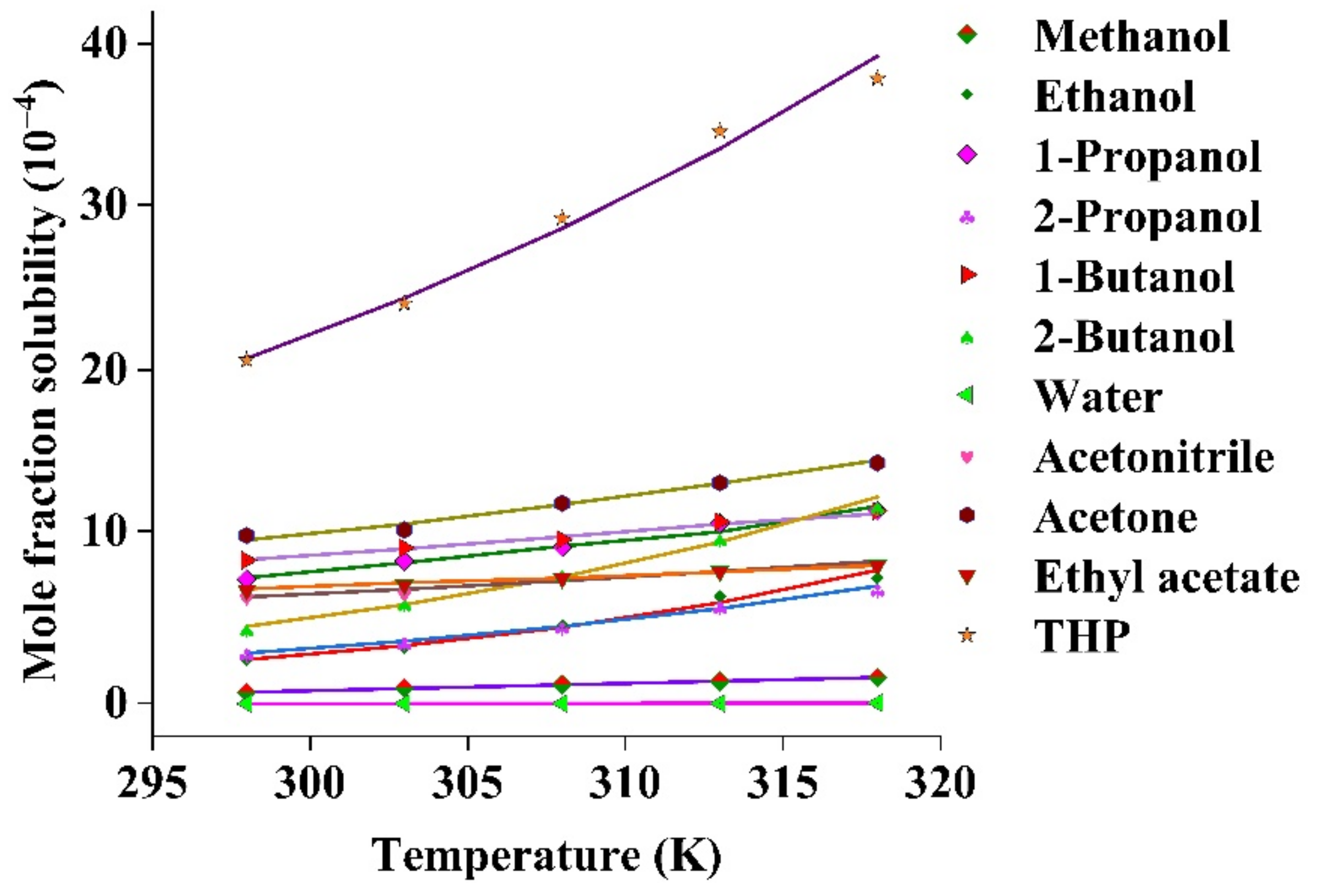
Organic_solvents Data With Water Solubility Solvent Functional Group
Common Organic Solvents_ Table of Properties Solvent Functional Group
The Solvent Is The Substance That Dissolves The Solute And The Component Of A Chemical Solution Present In The Greatest Amount.
A Solvent Is A Substance That Can Dissolve An Insoluble Solute And Create A Solution.
Solvent, Substance, Ordinarily A Liquid, In Which Other Materials Dissolve To Form A Solution.
Something That Provides A Solution.
Related Post: